Acids Bases Ph Worksheet Answers: A Comprehensive Guide
Understanding Acids and Bases
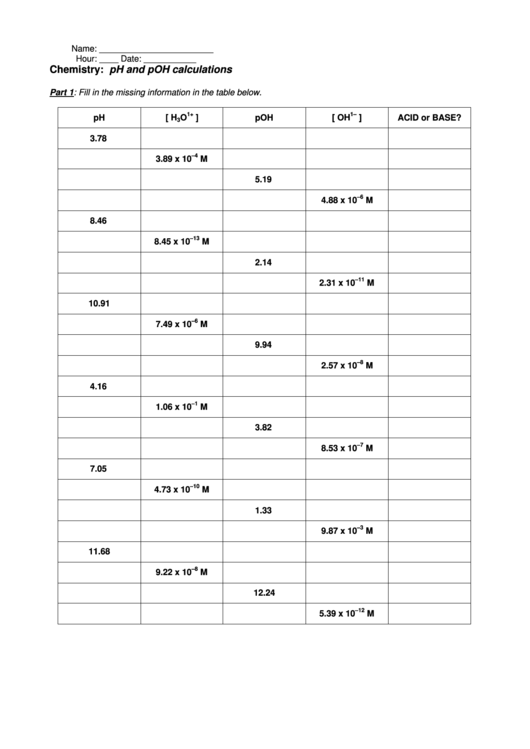
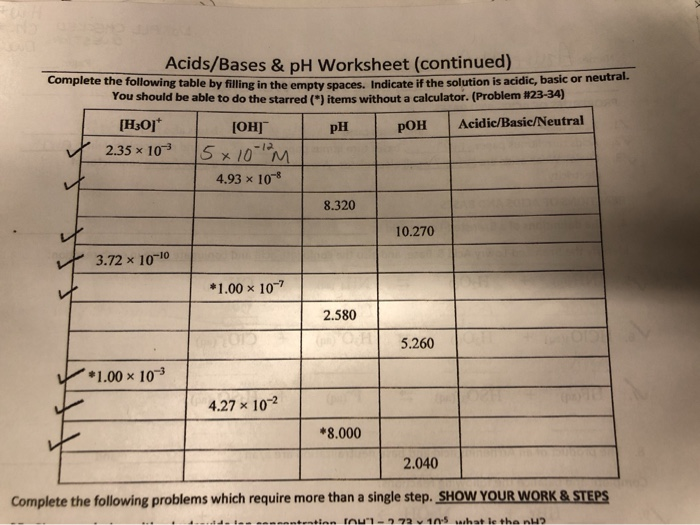
Acids and bases are fundamental concepts in chemistry, and understanding their properties and behavior is crucial for students and professionals alike. A pH worksheet is a great way to test your knowledge and skills in calculating the pH of different solutions. In this article, we will provide you with the answers to common acids and bases pH worksheet questions, as well as a brief overview of the key concepts.
When it comes to acids and bases, it's essential to understand the difference between strong and weak acids and bases. Strong acids and bases completely dissociate in water, while weak acids and bases only partially dissociate. This affects the pH of the solution, which is a measure of its acidity or basicity. The pH scale ranges from 0 to 14, with a pH of 7 being neutral.
Calculating pH: A Step-by-Step Approach
To calculate the pH of a solution, you need to know the concentration of hydrogen ions (H+) or hydroxide ions (OH-). The pH can be calculated using the formula pH = -log[H+], where [H+] is the concentration of hydrogen ions in moles per liter. For example, if the concentration of hydrogen ions is 0.01 M, the pH would be pH = -log(0.01) = 2. This means that the solution is acidic, with a pH of 2.
In conclusion, acids and bases pH worksheet answers can be found by understanding the basics of acids and bases, as well as the pH calculation formula. By practicing with sample problems and worksheets, you can improve your skills and become more confident in your ability to calculate pH. Whether you're a student or a professional, this guide provides a comprehensive overview of the key concepts and formulas you need to know to succeed in chemistry.